Clinical research blog
Explore our blog for insights into the big questions in precision medicine and clinical research.
As COVID-19 continues to affect us all, it has become evident that our fight against the virus is not just about developing vaccines and therapeutics. Equally c...
Continue reading
In this blog, we're exploring whether ALS can be caught early, including the tests that may be used in order to diagnose ALS, and if there are any measures that...
Continue reading
The success of any precision medicine program depends on the quality of the target it pursues. A well-validated genetic target shapes everything downstream: whi...
Continue reading
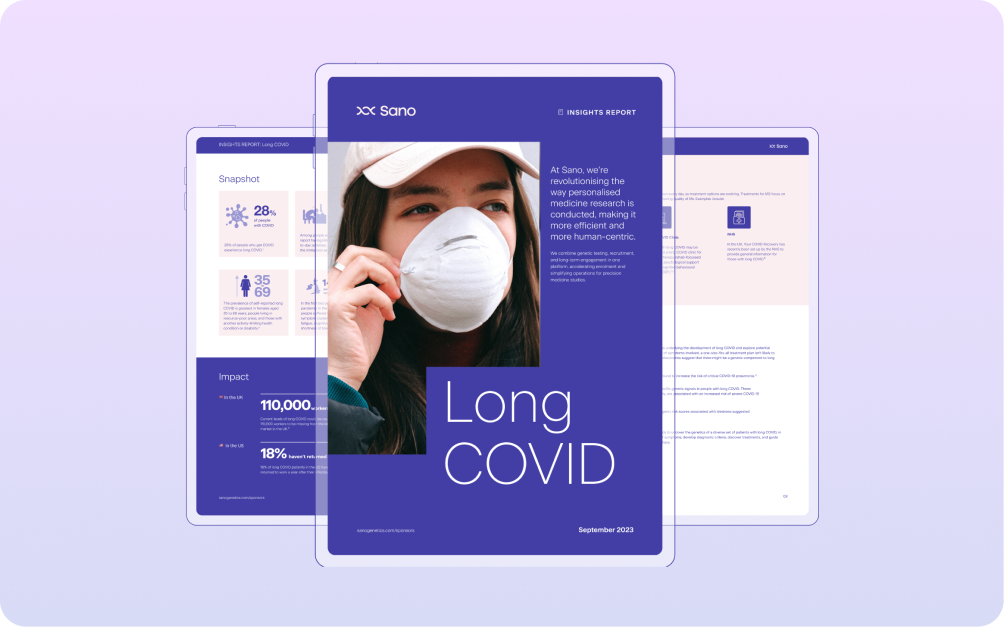
Long COVID, affecting nearly 28% of COVID-19 survivors, is a multifaceted condition that extends well beyond its initial impact. Typical include fatigue, concen...
Continue reading
In the ever-evolving landscape of clinical trial recruitment, an omnichannel approach is becoming increasingly crucial to ensure a diverse and engaged participa...
Continue reading
When an individual undergoes genetic testing, they often expect to find definitive answers about their genes. However, around 20% of genetic tests identify vari...
Continue reading
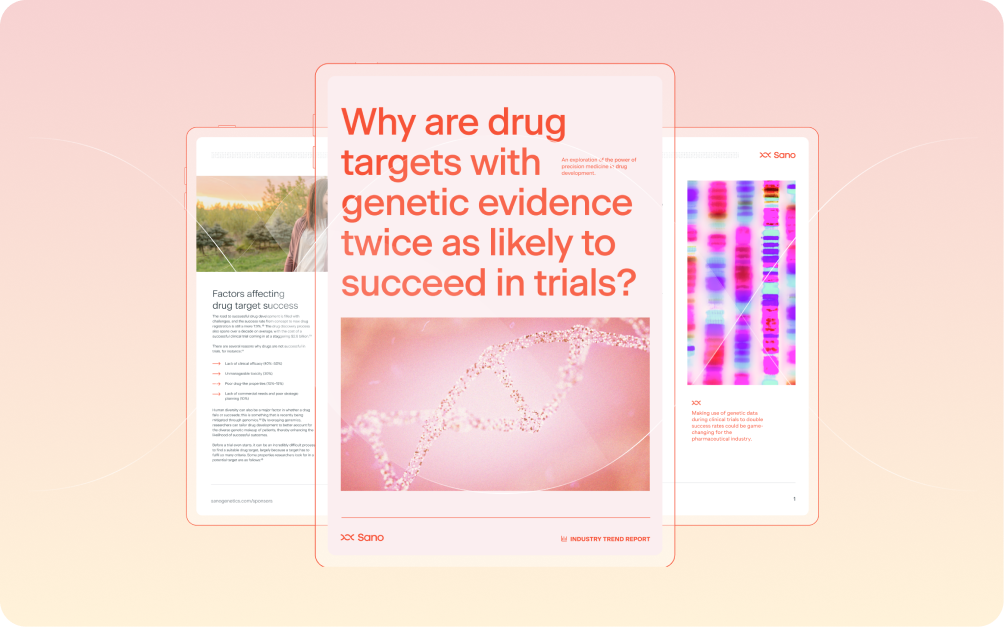
A 2015 study published in Nature found that by harnessing genetic data to support the selection of candidate drugs, researchers could double their success rates...
Continue reading
Clinical trials drive medical progress, but most are constrained by the same bottleneck: patient recruitment. Despite decades of investment in trial infrastruct...
Continue reading
In the ever-evolving world of healthcare and pharmaceuticals, researchers are constantly seeking ways to make drug development more efficient and effective. San...
Continue reading
Genetic data is now central to how drugs are discovered, developed, and brought to patients. For teams designing precision medicine programs, understanding wher...
Continue reading