Clinical research blog
Explore our blog for insights into the big questions in precision medicine and clinical research.
In the latest episode of The Genetics Podcast, Patrick speaks with Dr. Jim Wilson, Professor of Human Genetics at the University of Edinburgh, about his work st...
Continue reading
Sano is proud to support Neuron23 in the NEULARK Phase 2 clinical trial, a groundbreaking study exploring NEU-411, a brain-penetrant LRRK2 inhibitor with the po...
Continue reading
Last week on The Genetics Podcast, Patrick Short sat down with Michelle Teng, CEO and co-founder of Etcembly, to explore her work at the intersection of immunot...
Continue reading
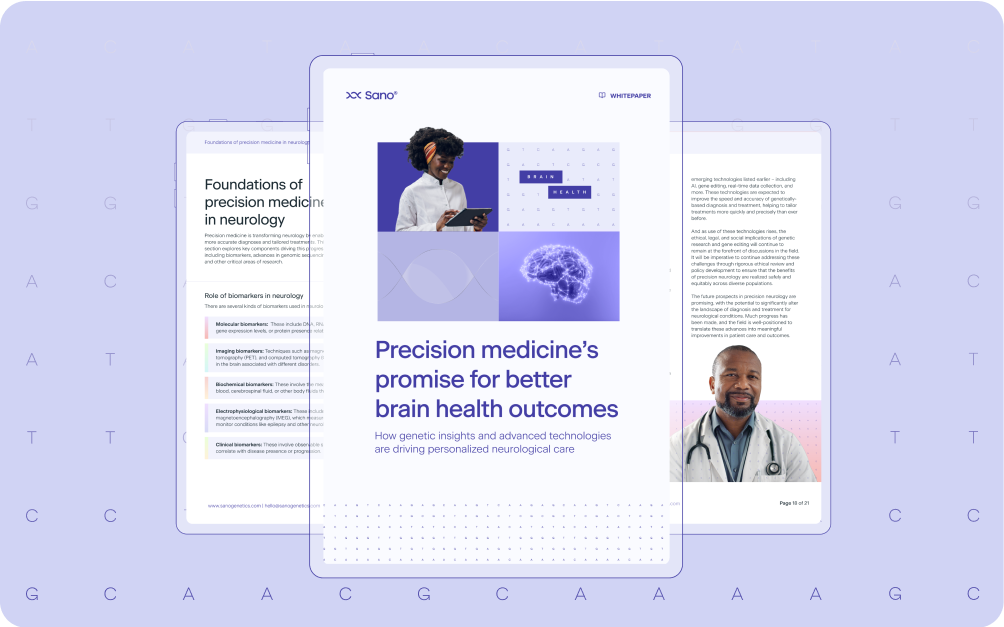
Precision medicine is transforming neurology by enabling more accurate diagnoses and tailored treatments. Here, we explore key components driving this progress,...
Continue reading
In a recent episode of The Genetics Podcast, host Patrick Short sits down with Eric Fauman, Executive Director and Head of Computational Biology in Pfizer's Int...
Continue reading
Millions of people worldwide are affected by neurological disorders like ALS, Alzheimer’s, and Parkinson’s. These conditions bring unique challenges for patient...
Continue reading
In the most recent episode of The Genetics Podcast, host Patrick Short speaks with Noam Baumatz, co-founder and CEO of Noga Therapeutics. Noam shares his deeply...
Continue reading
Today, the Lupus Research Alliance (LRA) launched the Data Repository, Exchange, and Analytics Platform (DREAM)—a pioneering knowledge-sharing platform dedicate...
Continue reading
In a recent webinar, former Reata Pharmaceuticals CEO Warren Huff shared his journey leading the company from its founding in 2002 to its landmark acquisition b...
Continue reading
Inherited retinal disorders (IRDs) are a diverse group of genetic eye conditions that impact millions worldwide, affecting light-sensitive cells in the retina a...
Continue reading