Key Takeaways
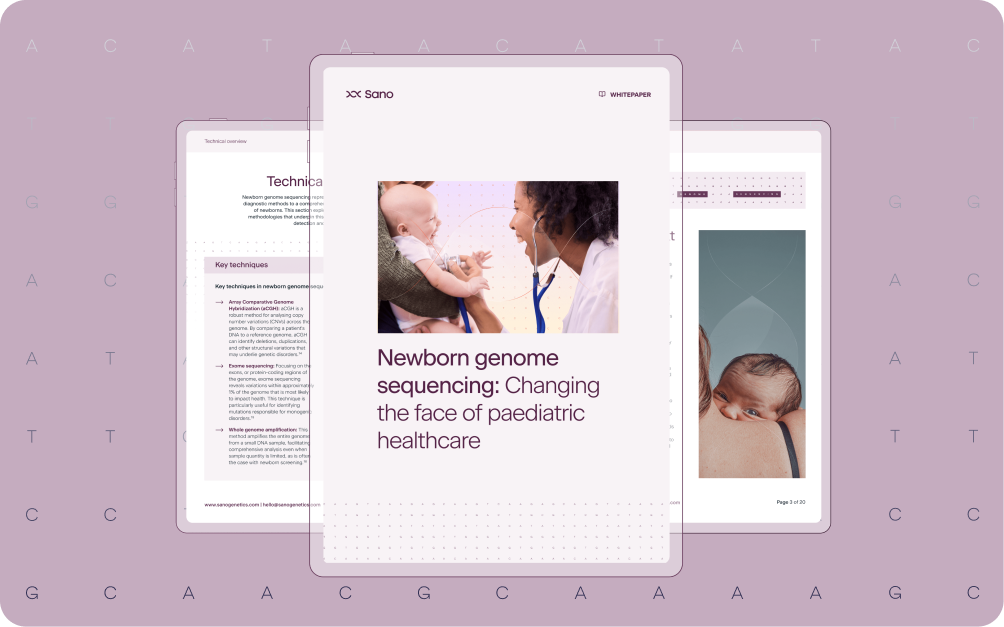
- Newborn genome sequencing identifies a broader spectrum of genetic disorders than traditional heel prick tests.
- Early detection enables highly personalized healthcare strategies from the start of life.
- Integration requires addressing critical ethical concerns regarding privacy, consent, and equitable access.
- The combination of AI and genomics is expected to significantly refine screening accuracy.
Navigating challenges and looking ahead
While the potential benefits are substantial, the adoption of genome sequencing in newborn screening introduces real challenges that must be addressed carefully. The risk of false positive results is one. Each healthy human genome contains hundreds of gene changes that appear severe on paper, and interpreting these variants in asymptomatic infants remains difficult. Not every baby flagged as "at risk" will go on to develop a condition, which raises questions about how to communicate uncertain findings to parents without causing unnecessary anxiety.
Beyond clinical interpretation, there are unresolved questions about consent, data storage, and the secondary use of genomic information over a lifetime. Programs like the Generation Study are actively engaging with parents, rare disease communities, and policymakers to examine these implications. Our whitepaper explores these issues in depth, proposing frameworks for integrating new genetic technologies in a manner that respects both individual rights and public health needs.
One of the structural advantages of genome sequencing is its expandability. Once a newborn's genome is sequenced, the data can be re-analyzed as new conditions are characterized and new interventions become available. This means the screening panel is not fixed at the point of collection. It can grow alongside therapeutic development. For sponsors and researchers working in precision medicine, this creates a fundamentally different relationship between early genomic data and long-term clinical utility.
Newborn genome sequencing is not only reshaping pediatric care. It is creating new populations of genetically characterized individuals who may become relevant to future precision medicine programs. For a detailed examination of the clinical, ethical, and operational dimensions of this shift, download the whitepaper.